ISO 9001:2015 VS ISO 13485:2016 MATRIX HOW TO
Non-Conforming Process – AS9100 has more specific requirements on how to handle non-conformances and how to take action on processes and products, including what corrective actions need to take place if a nonconformance is found.The changes include production processes, as well as required controls on production equipment and support after delivery.
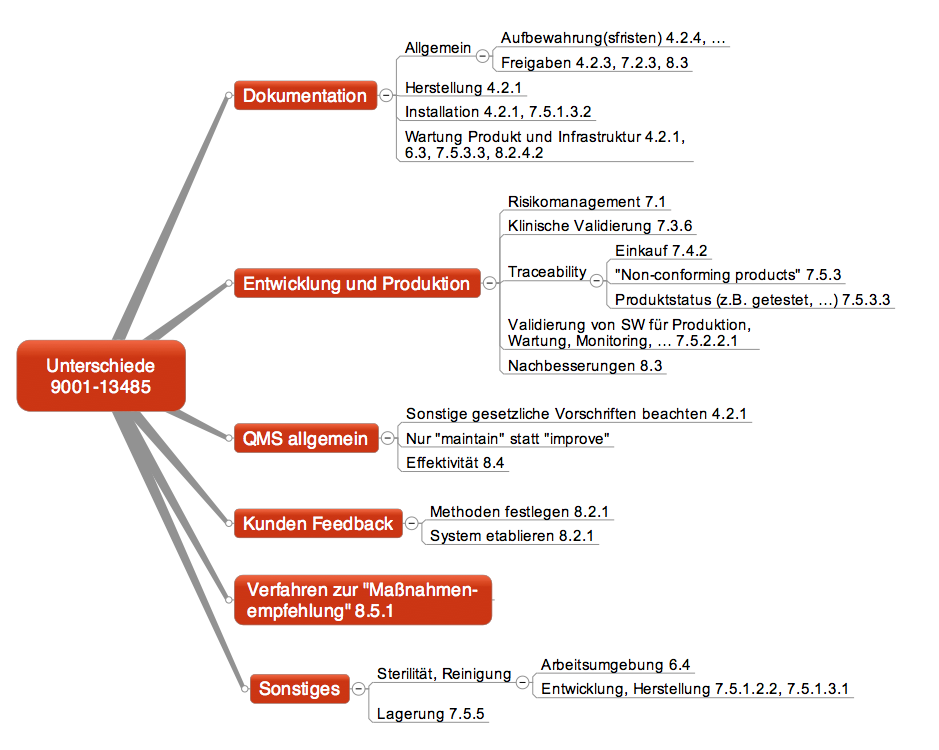
ISO 9001:2015 VS ISO 13485:2016 MATRIX MANUAL
The reason is that this is the biggest difference from one industry to the other. Correspondence Between ISO 13485:2016 and 21 CFR 820 Regulatory Compliance Associates® Inc., 10411 Corporate Drive, Suite 102, Pleasant Prairie, WI 53158 5:2016 US FDA Quality System Regulation (QSR - 21 CFR 820) The quality manual shall outline the structure of the documentation used in the quality management system. Production and Service Provision – This topic has the most changes in AS9100.Purchasing and Purchased Product – This section contains many additional requirements regarding supplier control.Risk identification and assessment are carried throughout the requirements of the standard as risk is critical for the aerospace industries.

